Hi online CHEM 151 students! I hope your wrap up of Unit 1 is going well, as well as your final prep for the Unit 1 exam (if you haven’t taken it already). The following is a list of reminders for the coming week as you wrap up Unit 1 and begin Unit 2.
Unit 1 Exam
If you haven’t already taken the Unit 1 exam, please see the info at the end of this page under Unit 1 Exam
Unit 2
The next unit is Unit 2 and it will move more into the study of chemistry, as opposed to the calculational tools emphasized in Unit 1 (especially in the Matter and measurement section). An overview of Unit 2 is provided at this link: Unit 2.
The organization of Unit 2 is similar to Unit 1 – the assignments and deadlines for Unit 2 can be seen on the Online Course Schedule – the next deadline (and the first for this Unit) is Sept 21.
Online Homework
The online homework for all three assignments in Unit 1 is due on Monday, Sept 11.
For those of you doing the honor’s homework (or interested in it), it is being developed this semester and I’m a little behind on that (my apologies). The first assignment is available now (Essential ideas – matter and measurement). The other two (Atoms, molesules and ions and Stoichiometry) will be available over the weekend. Since they have not been available yet, they can be completed anytime before the end of September.
Please contact me if you have interest in or questions about the Honors option.
Quiz/Class Participations
Quiz 3’s have been graded and returned to you. There was an error in question 3. It said “How many oxygen atoms are in 1500 atoms of C6H12O6.” It should have said “How many oxygen atoms are in 1500 molecules of C6H12O6.” Because of this error, you got full credit regardless of your answer.
The correct answer to the question as it should have been written is:
1500 molecules * 6 O atoms/molecule = 9000 O atoms.
Also, question 2 on Quiz 3 asked for the number of protons, neutrons, and electrons in an iron-58 atom. Several of you answered 26 protons (correct), 32 neutrons (correct) and 28 electrons (incorrect). Some of you explained that it was Fe2-. An Fe atom is neutral (zero charge) by definition. Only an ion has a charge. In a neutral atom, the number of protons and electrons are the same, so the correct number of electrons is 26. So why was 28 electrons a common (incorrect) answer? I suspect it was a result of a Google search like this: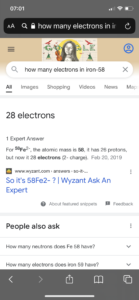
This definitely looks like the likely source of the error. I’m not going to tell you not to google to try to find chemistry help or answers — for better and worse it’s how we get a lot of our information these days, and you probably wouldn’t listen to me anyway :). I will point out some problems/cautions with this approach:
- No doubt google searches can be helpful, but you have to dig a little deeper, read the links the google search gives you and consider the question you are being asked as well as the source writing it. Clicking on the link or reading below said there were 28 electrons in Fe2-. The quiz question wasn’t about an Fe2- ion, it was about an Fe atom.
- Google searches do not always save you time. If you understood that in a neutral atom the number of protons equals the number of electrons and that the atomic number is the number of protons, a quick look at a periodic table would tell you that an iron atom has 26 protons (element 26) and therefore 26 electrons, This would have been faster than a Google search.
It will be helpful for you to think about this as you choose how much to incorporate (or not) google searches into your studies (in addition to looking through the provided material) as you progress through the course.
Unit 1 Exam
Exam Information
The exam is a proctored, pen/pencil and paper exam. It is not offered online on D2L. It will be available during open Testing Center Days and Hours ending Monday 9/11.
The exam will cover:
Testing hours and locations are at this link
There is detailed information about the exams at this link. Please read it carefully – it contains the answers to many frequently asked questions. Let me know if you have additional questions after reading it.
For this exam, you will be provided a copy of the Periodic Table for Units 1-3. A copy of the periodic table you will receive is at this link.
Tutoring and Help Opportunities from the Learning Commons
The link to the Learning Commons Chemistry regular tutoring schedule and special exam prep schedule is here (and on the right sidebar of this website)
Learning Commons Chemistry Tutoring Schedule
CHEM 151 Exam Prep sessions hosted by LCC tutoring
Additional Study Resources
Don’t forget to look at the Unit 1 – additional study resourcespage on this Website. It contains many resources to help you prepare for the exam. Please take a look at them. One of those resources is a practice exam. Please read more about the practice exam below:
Practice Exam
What the practice exam is:
This practice exam will be similar in length, number of questions, types of questions (multiple choice, short answer, numerical problems, etc) to your exam. The practice exam will be useful as a general review and will help you see how prepared you are. If the practice exam (without looking at your notes) is easy for you, the actual exam probably will be. If it is difficult, the actual exam probably will be.
What the practice exam is NOT:
The actual exam will NOT be the same as the practice exam with only a few numbers changed. Do NOT think that if you only study and memorize the questions on the practice exam that you will be ready for the exam because the questions will be the same.
